|
12/18/2023 0 Comments 60 carbon atoms 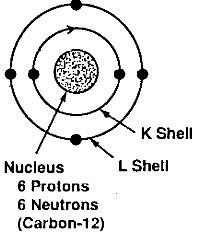
Manuel lvarez Bravo es un_ famoso de la UNAM. 4.Los_ de la UNAM son de diferentes pases. 3.La UNAM es como (like) una ciudad con_, polica y gobierno (government) propios (own). _es un importante centro econmico y cultural. fill in the blanks with the appropriate words from the list.-Mxico, D.F.autobusesderechoestudiantesestudioexalumnoexprofesorresidencia estudiantilUniversidad1. I need help, I thought I was placing the right word in the correct spots, but I'm not.What makes America great ? Explain with reason.They have almost a horizontal slope where the particles are deposited by both wind and water.ĭetails : Identify landform D, explain how it forms, and tell whether it is In the image given above, landform D is most probably a pediplain area formed due to the accumulation of sediments away from the region of alluvial fans. Lastly, some of the depositional features formed by ice are mostly the different types of moraines, outwash plains, and drumlins. The depositional features formed by water are the beaches, deltas, and sand dunes. Some of the depositional landforms formed by wind are barchans and seifs. These agents when moves or flows over a surface, carry the sediments and deposits at a different place, creating various types of landforms. This process generally takes by agents such as wind, water, and ice. These features are formed due to the accumulation of the materials and the sediments. The landform D that is represented in the given image depicts a depositional feature. So volume, moles and mass are all directly proportional to each other \frac Īs volume increases, the number of moles occupies too will increase. When there is a change in volume or number of moles we make use of the expression Where V is the volume, n is the number of moles and k represents a constant. V\propto n (at constant temperature and pressure) That is, Equal volume of different gases will have the same number of moles at constant temperature and pressure.ġ mole of Hydrogen gas at T = 273K and P = 1 atm occupies a volume of 22.4Lġ mole of Argon gas at T = 273K and P = 1 atm occupies a volume of 22.4L Volume is the space occupied by a substance.Īt constant temperature and pressure, the volume occupied by a gas is directly proportional to the number of moles of the gas.ĭirectly proportional means when one increases the other one too will increase. Mass is the amount of matter present in a substance So we see here volume is directly proportional to its mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
